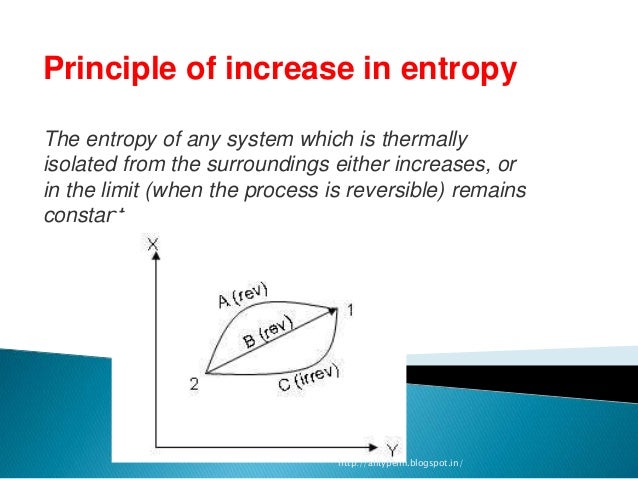
Our results cast doubt on the possibility of defining a state function that plays the role of a thermodynamic entropy for nonequilibrium steady states. In other words, a nonequilibrium steady state is not determined by maximizing the Shannon entropy. Entropy (S) is a state function that can be related to the number of microstates for a system (the number of ways the system can be arranged) and to the. The inequivalence of S S and S t h implies that derivatives of S S are not predictive of coexistence. We verify that S t h is not a state function: changes Δ S t h depend not only on the initial and final points, but also on the path in parameter space. Although the two entropies are identical in equilibrium (as expected), they differ out of equilibrium for small values of the drive, D, we find | S S − S t h | ∝ D 2, as expected on the basis of symmetry. A state function is a property depending only on the. We determine via direct calculation the stationary nonequilibrium probability distribution of the driven lattice gas with nearest-neighbor exclusion, the Katz-Lebowitz-Spohn driven lattice gas, and a two-temperature Ising model so that we may evaluate the Shannon entropy S S as well as S t h defined above. The two processes have the same initial and final states, therefore, the same S. In classical thermodynamics the entropy is interpreted as a state function of a thermodynamic system. In effect 1/ T is an integrating factor which, when it multiplies the inexact differential Q, results in the exact differential Q / T dS. These results suggest that an associated steady-state entropy S t h be constructed via thermodynamic integration, using relations such as ( ∂ S / ∂ E ) V, N = 1 / T, ensuring that derivatives of S t h with respect to energy and particle number yield the expected intensive parameters. This means that the net change in entropy during a complete cycle is zero, so that entropy is a function of state. For an appropriate choice of exchange rates, T and μ satisfy the zeroth law, marking an important step in the development of steady-state thermodynamics. 
For stochastic lattice models in spatially uniform nonequilibrium steady states, definitions of temperature T and chemical potential μ have been verified using coexistence with heat and particle reservoirs. I have written the Clausius-Duhem inequality in the form of rates that it is true no matter how fast the process is but you can equally well write it as $d\mathcal E \le TdS+\delta \mathcal W$ since both internal energy and entropy are state functions, hence the $d$, but since work is not we need the $\delta$ to emphasize it.Defining an entropy function out of equilibrium is an outstanding challenge. The term and the idea are utilized in assorted fields. Entropy is a logical idea as well as a quantifiable actual property that is generally ordinarily connected with a condition or disorder, randomness, or vulnerability. Entropy is characterized as the degree of randomness or proportion of turmoil. 
There is no physical process by which one may increase the internal energy *faster* than is allowed by this so-called Clausius-Duhem inequality. Introduction of entropy as a state function. Gibbs free energy of the system is a state function because it is defined in terms of thermodynamic properties that are state functions. This inequality shows how fast you can increase the internal energy by working on the body. State Functions also know as point functions are defined as a property whose value doesn’t depend on the path taken to reach that specific value. However, if you do not make the substitution you do not get zero so the two. During isothermal and reversible expansion of the gas from volume v(1) to v(2), let there be absorption of heat q at temperature T. The proof requires a substitution of p T n R V because when it is then differentiated with respect to T it equates to zero and so does C V T when it is differentiated with respect to V which shows it is an exact differential. e., the temperature, pressure, volume, etc The value of a state function is independent of the history of the. Consider a cylinder fitted with a frictionless and weightless piston, which contains a gas and is in contact with a large heat reservoir. A State Function is a thermodynamic quantity whose value depends only on the state at the moment, i. Between two equilibrium states, say $\mathcal $ is the rate of working (i.e., "power") done on the body whose internal energy is $\mathcal E$. State function is a quantity in thermodynamics, such as entropy or enthalpy, that has a unique value for each given state of a system. Where $\delta Q$ is the heat absorbed at temperature $T$ note that the entropy does not come in to play here. The Clausius inequality states that over any thermodynamic cycle
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
